McDermott+ is pleased to bring you Regs & Eggs, a weekly Regulatory Affairs blog by Jeffrey…
Continue ReadingCategory: Digital Health and Medical Technology
Custom Pain Management Services for Auto Injury
Recovering from an auto accident is often more complicated than people expect. Many patients feel “mostly…
Continue Reading
New Semiconductor Chip Developed for Portable Medical Devices Enables Advanced Self-Diagnostics
• 1 day ago • 3 Mins Read The IISc-developed chip performs real-time biomedical signal processing…
Continue Reading
FDA launches digital health pilot centred on chronic disease
The FDA’s TEMPO pilot will begin in January 2026. image credit: Photo Agency via Shutterstock.com. The…
Continue Reading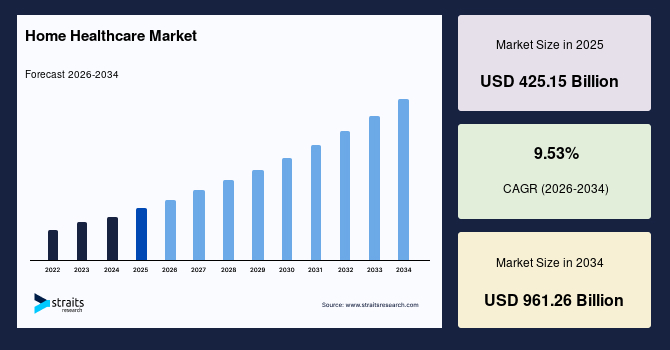
Home Healthcare Market Size, Share & Growth Report by 2034
Home Healthcare Market Overview The global home healthcare market size is valued at USD 425.15 billion…
Continue Reading
New EU4Health projects push digital health forward across Europe
image: ©BlackJack3D | iStock A new wave of EU-funded initiatives launched under the 2024 EU4Health Work…
Continue Reading
Q&A: Real-time digital healthcare for 2026
A radiologist interpreting magnetic resonance imaging. Image by The Medical Futurist editors. – The Future of…
Continue Reading
S3 Connected Health receives Frost & Sullivan 2025 Global Company of the Year award
S3 Connected Health has received Frost & Sullivan’s 2025 Global Company of the Year award for…
Continue Reading
UK reforms highlight the case for digital submissions in medical device regulation
The UK’s Medicines and Healthcare products Regulatory Agency’s (MHRA) reforms emphasise digital post-market surveillance, interoperability with…
Continue Reading
U.S. Software as a Medical Device Market to Reach USD 715 Million by 2033 as AI-Driven Diagnostics and Digital Therapeutics Accelerate Adoption
Classified in: Health, Science and technologySubject: MRR AUSTIN, Texas and TOKYO, Nov. 24, 2025 /PRNewswire/ —…
Continue Reading
